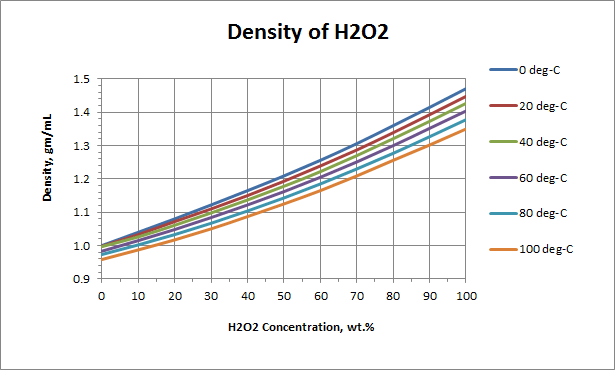
SOLVED: 1,100 kgIm' Whal would you expect the mass ofthat solution to be if it 309 hydrogen peroxide has density of filled container with volume of 0,03 m ? 990 kg 33kg 0000027 kg 36,666.67 kg

OneClass: If I have hydrogen peroxide, molecular weight(g/mol) is 34.02, its density (g/ml) is 1.2, i...

Table 1 from Biodecomposition of Hydrogen Peroxide (H 2 O 2 ) in Water and in Organic Solvents Using Saccharomyces cerevisiae Meyen ex E.C. Hansen (Fungi: Ascomycota) | Semantic Scholar

SOLVED: You are given an aqueous solution of hydrogen peroxide that is 30.0% by mass and has a density of 1.11 g/mL What is the molality of the solution? Molar mass of

Concentration of Hydrogen Peroxide in a 10 Volume Solution - An Interesting Stoichiometry Problem - YouTube
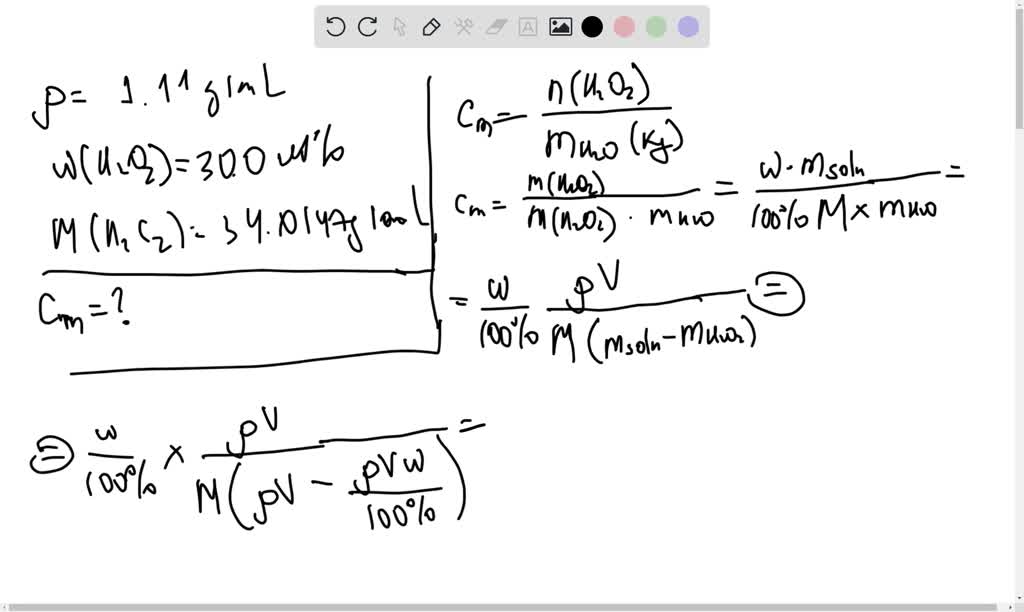
SOLVED: Aqueous solutions (density 1.11 g/mL) of 30.0 wt% hydrogen peroxide and H2O2 (molar mass 34.0147 g/mol) are used to oxidize metals or organic molecules in chemical reactions. Calculate the molality in