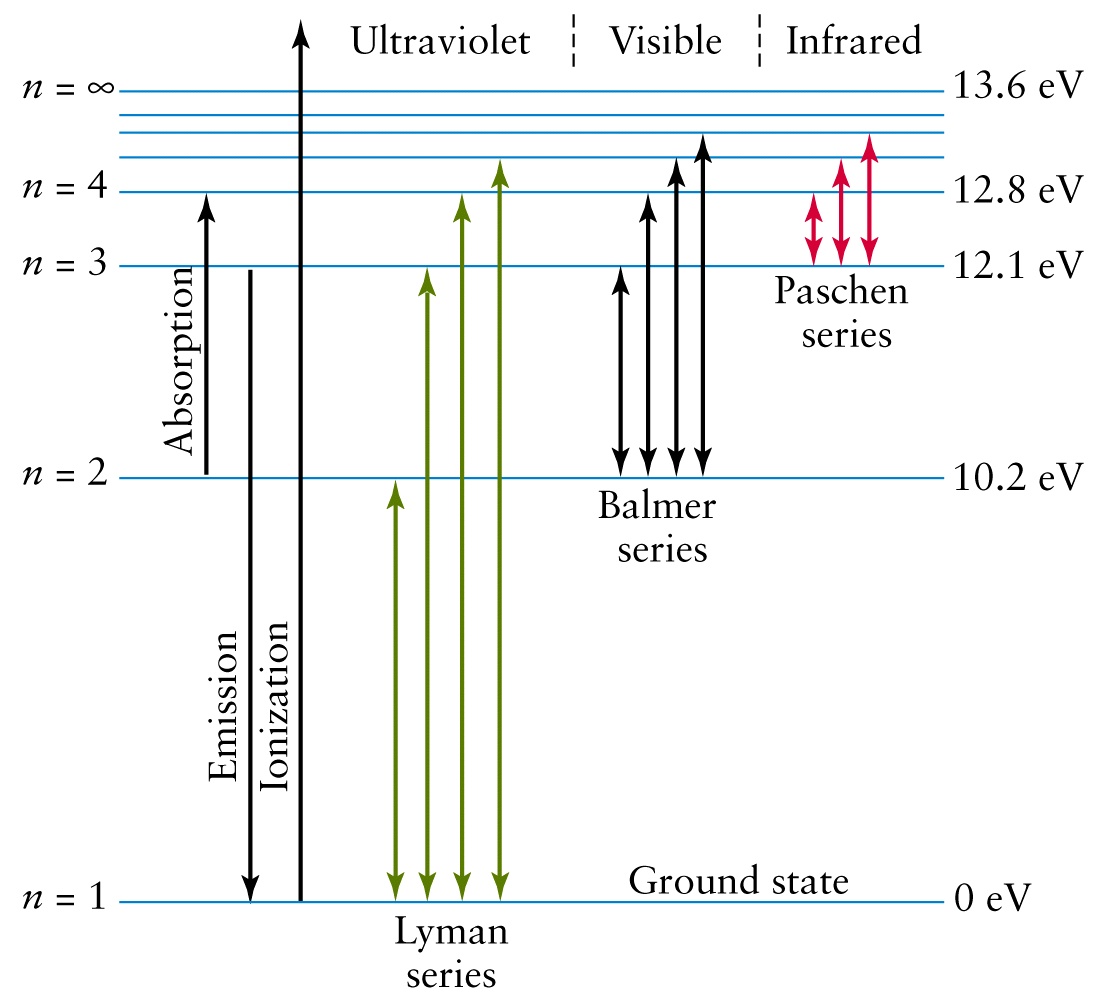
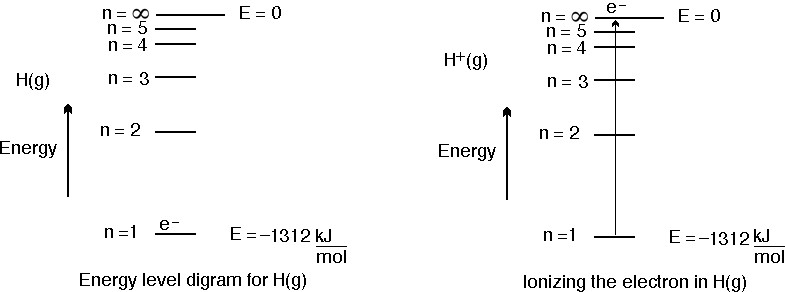
Calculate the energy required for the process , He^+ (g) rarr He^(2+) (g) + e The ionization energy for the H-atom in the grounds state is 2. 18 xx 10 ^(-18) J "atom"^(-1).

Super Trick to Find Ionization Energy in 20 Seconds || How to Find Ionization Energy || Chemistry - YouTube

OneClass: Please show all work 5. Use the following equation to calculate the ionization energy in el...

SOLVED: 20. The first ionization energy (IE1) is the amount of energy required to move the electron out of the atom completely (from n - to n = in the Bohr equation)
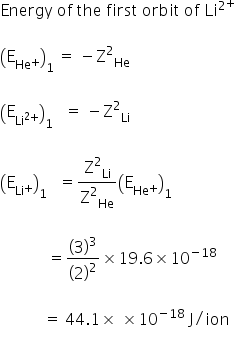
the ionization enery of he is 196 10 18 j ioncalculate the enery of the first stationary state of li2 24b6v955 -Chemistry - TopperLearning.com