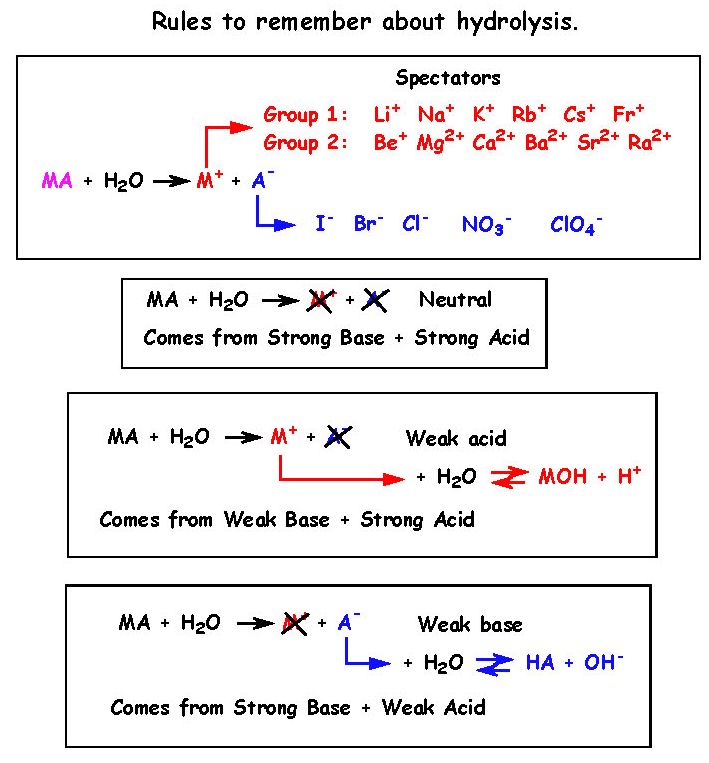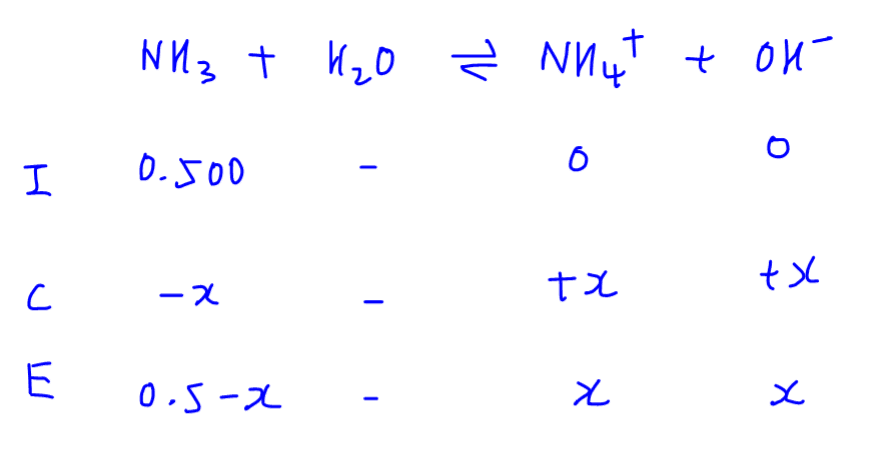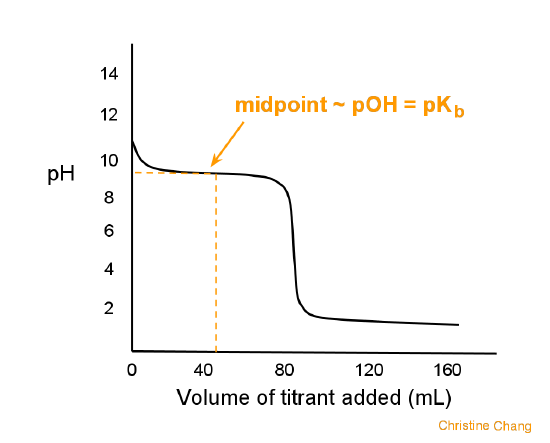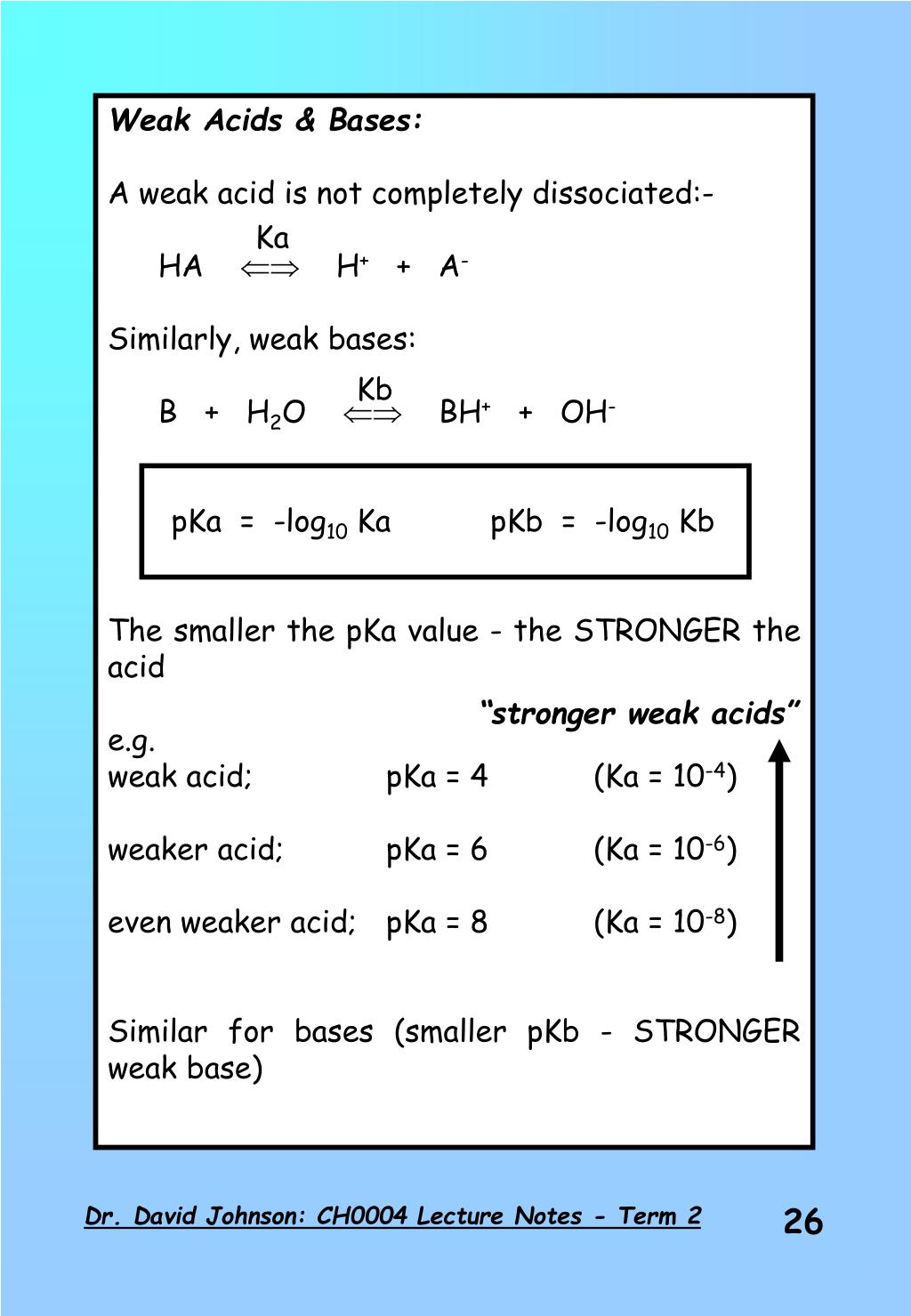
PPT - Weak Acids & Bases: A weak acid is not completely dissociated:- HA H + + A - PowerPoint Presentation - ID:3428717

SOLVED: Amphetamine (C9H13N) is a weak base with a pKb of 4.2. Calculate the pH of a solution containing an amphetamine concentration of 225 mg>L.
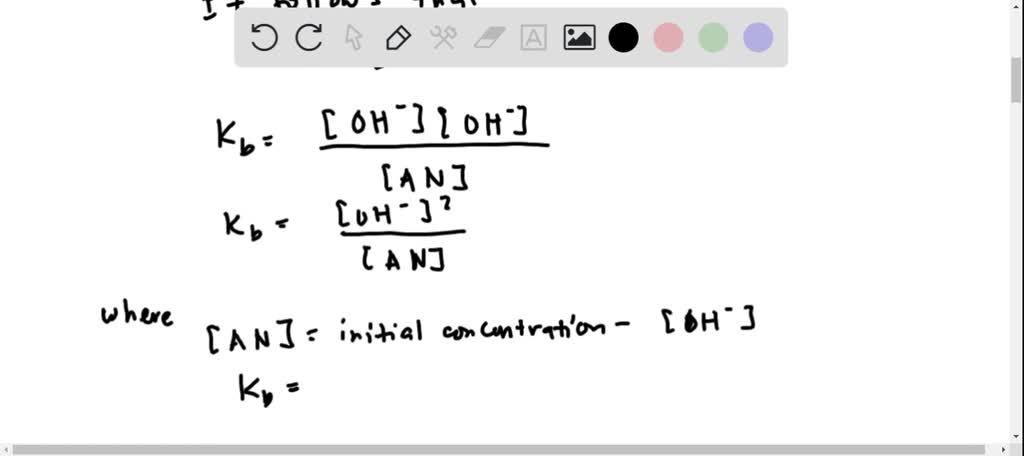
SOLVED: Amphetamine (C9H13N) is a weak base with a pKb of 4.2. Calculate the pH of a solution containing an amphetamine concentration of 225 mg>L.
The pK a of a weak acid (HA) and pKb of weak base (BOH) are 3.2 and 3.4, respectively. The pH of their salt (AB) solution is - Sarthaks eConnect | Largest Online Education Community
![SOLVED: Usefulequations: pH pKa + log ([conjV[WA]) where WA = weak acid pOH = pKb + log ([conjV[WB]) where WB weak base pH = -log1o[HgOt]; [H3Ot] = 10- pH pOH = log10[OH-]; [OH:] = SOLVED: Usefulequations: pH pKa + log ([conjV[WA]) where WA = weak acid pOH = pKb + log ([conjV[WB]) where WB weak base pH = -log1o[HgOt]; [H3Ot] = 10- pH pOH = log10[OH-]; [OH:] =](https://cdn.numerade.com/ask_images/614c9971ddeb4795b21a29f9ef72aa78.jpg)
SOLVED: Usefulequations: pH pKa + log ([conjV[WA]) where WA = weak acid pOH = pKb + log ([conjV[WB]) where WB weak base pH = -log1o[HgOt]; [H3Ot] = 10- pH pOH = log10[OH-]; [OH:] =
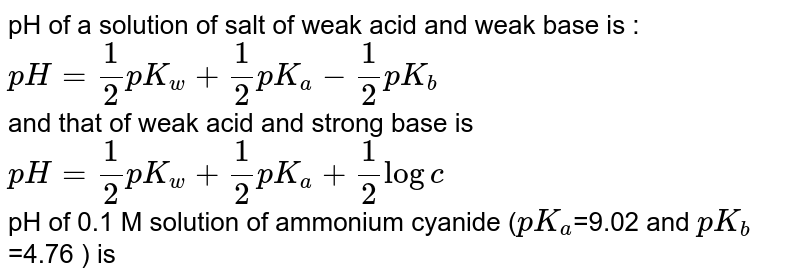
pH of a solution of salt of weak acid and weak base is : pH=1/2pKw+1/2pKa- pKb and that of weak acid and strong base is pH=1/2pKw+1/2pKa+1/2logc The pH of 0.1 M sodium acetate (

pH of a solution of salt of weak acid and weak base is : pH=1/2pKw+1/2pKa- pKb and that of weak acid and strong base is pH=1/2pKw+1/2pKa+1/2logc The pH of 0.1 M sodium acetate (