Section 2 - Avogadro's Number and the Mole - Part 1 | Math Tutor DVD - Online Math Help, Math Homework Help, Math Problems, Math Practice!

Physical Chemistry #1: Relative Mass, the Mole and Avogadro's Constant (Slides & Student Led Tasks) | Teaching Resources

Calculate the value of Avogadro number from the internuclear distance of adjacent ions in NaCl , 0.282 nm and the density of solid NaCl is 2.17 × 10^3 kg/m^3 .A unit cell

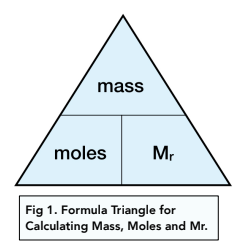
The Mole Formula Triangle Or Pyramid With Avogadro Number Or Avogadro Constant Isolated On White Chemistry Stock Illustration - Download Image Now - iStock
![How to calculate number of particles| Avogadro's constant| Mole and Chemical Calculation [Online Video] – O Level Secondary Chemistry Tuition How to calculate number of particles| Avogadro's constant| Mole and Chemical Calculation [Online Video] – O Level Secondary Chemistry Tuition](https://icandochemistry942105908.files.wordpress.com/2020/12/thumb-insta-2.jpg?w=640)
How to calculate number of particles| Avogadro's constant| Mole and Chemical Calculation [Online Video] – O Level Secondary Chemistry Tuition

SOLVED: Use your data to calculate the uncertainty in: (a) your value of Avogadro's number number of molecules of SA N * M NA number of moles of SA m %U (NA) = %